Contents
© The scientific sentence. 2010
| Spectral line emissions series
0. Rydberg formula
 Eni = E = - 13.6 (Z2/ni2) eV
Enf = E = - 13.6 (Z2/nf2) eV
ΔE = Enf - Eni =
- 13.6 (Z2/nf2) + 13.6 (Z2/ni2) =
13.6 Z2 (1/ni2 - 1/nf2)
ΔE = h ν = hc/λ
Therefore:
hc/λ = 13.6 Z2 (1/ni2 - 1/nf2)
Or:
1/λ = (13.6/hc)Z2 (1/ni2 - 1/nf2)
h = 6.62606957(29)×10-34
c = 3.0 x 108 m/s
13.6 eV = 13.6 . 1.6 x 10-19 J
(13.6/hc) = 13.6 . 1.6 x 10-19/6.63 x 10-343.0 x 108 =
1.10 x 10 +34 - 8 - 19 = 1.10 x 107 (1/m)
1/λ = 1.10 x 107 Z2 (1/ni2 - 1/nf2 ) (1/m)
Using the Rydberg constant:
Ry = 1 Rydberg = 1.0974 x 107 m-1, we have:
1/λ = Ry Z2 (1/ni2 - 1/nf2 )
Therefore:
λ = 0.91 x 10- 7/Z2[1/(1/ni2 - 1/nf2 )] (m)
λ = 91.1 /Z2(1/ni2 - 1/nf2 )] (nm)
For any layer n, its energy En is less than
the one of its next En+1. Then if the transition
goes from ni = n+1 toward nf = n , the enrgiy difference ΔE is
GIVEN, and still NEGATIVE. Thus, in order
to have a positive wavelength, we just solve
the following formula by taking the positive value (or
absolute value)for &lamda;, or invert the order of nf
and ni, so:
λ = (91.1/Z2) /(1/nf2 - 1/ni2 )] (nm)
This formula is valid for a hydrogen atom
and hydrogen-like ion(any atomic nucleus with
one electron). For atom with many electros, we
use instead Moseley's law
.
Eni = E = - 13.6 (Z2/ni2) eV
Enf = E = - 13.6 (Z2/nf2) eV
ΔE = Enf - Eni =
- 13.6 (Z2/nf2) + 13.6 (Z2/ni2) =
13.6 Z2 (1/ni2 - 1/nf2)
ΔE = h ν = hc/λ
Therefore:
hc/λ = 13.6 Z2 (1/ni2 - 1/nf2)
Or:
1/λ = (13.6/hc)Z2 (1/ni2 - 1/nf2)
h = 6.62606957(29)×10-34
c = 3.0 x 108 m/s
13.6 eV = 13.6 . 1.6 x 10-19 J
(13.6/hc) = 13.6 . 1.6 x 10-19/6.63 x 10-343.0 x 108 =
1.10 x 10 +34 - 8 - 19 = 1.10 x 107 (1/m)
1/λ = 1.10 x 107 Z2 (1/ni2 - 1/nf2 ) (1/m)
Using the Rydberg constant:
Ry = 1 Rydberg = 1.0974 x 107 m-1, we have:
1/λ = Ry Z2 (1/ni2 - 1/nf2 )
Therefore:
λ = 0.91 x 10- 7/Z2[1/(1/ni2 - 1/nf2 )] (m)
λ = 91.1 /Z2(1/ni2 - 1/nf2 )] (nm)
For any layer n, its energy En is less than
the one of its next En+1. Then if the transition
goes from ni = n+1 toward nf = n , the enrgiy difference ΔE is
GIVEN, and still NEGATIVE. Thus, in order
to have a positive wavelength, we just solve
the following formula by taking the positive value (or
absolute value)for &lamda;, or invert the order of nf
and ni, so:
λ = (91.1/Z2) /(1/nf2 - 1/ni2 )] (nm)
This formula is valid for a hydrogen atom
and hydrogen-like ion(any atomic nucleus with
one electron). For atom with many electros, we
use instead Moseley's law
.
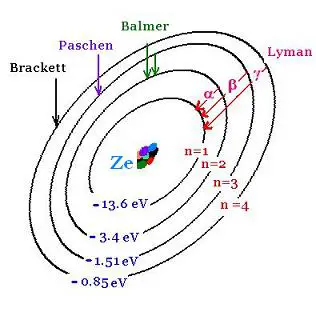
1. Lyman series: → nf = 1
Lyman series are the series of transitions for an atom
when one of its electrons goes from a layer n to the layer
n = 1 . The number n is the principal quantum number
(corresponding to a certain level of enrgy of an electron
in the atom.
The transitions:
from n = 2 to n = 1 is called Lyman-alpha,
from n = 3 to n = 1 is Lyman-beta,
4 to 1 is Lyman-gamma, etc.
All these transitions are in the range of
the ultraviolet.
We have the Rydberg formula:
1/λ (n) = Ry(1/12 - 1/n2)
Ry = 1 Rydberg = 1.0974 x 107 m-1
or:
λ = (1/Ry) . 1/(1 - 1/n2)
= 91.1 /(1 - 1/n2) nm
Lyman-alpha:
λ (2) = 91.1/(1 - 1/22) = 121 nm
Lyman-beta:
λ = 91.1/(1 - 1/32) = 102 nm
Lyman-gamma:
λ = 91.1/(1 - 1/42) = 97 nm
2. Balmer series: → nf = 2
Balmer series are the series of transitions for an atom
when one of its electrons goes from a layer n to the layer
n = 2.
The transitions:
from n = 3 to n = 2 is called Balmer-alpha,
from n = 4 to n = 2 is Balmer-beta,
5 to 2 is Balmer-gamma,
6 to 2 is Balmer-delta, etc.
All these transitions are in the range of
the visible. For n>7 →2 the
spectrum is the ultraviolet.
We have the Rydberg formula:
λ = (1/Ry) . 1/(1/22 - 1/ni2)
= 91.1 /(1/22 - 1/n2) nm
Balmer-alpha:
λ (2) = 91.1/(1/4 - 1/32) = 656 nm (red)
Balmer-beta:
λ = 91.1/(1/4 - 1/42) = 486 nm (cyan)
Balmer-gamma:
λ = 91.1/(1/4 - 1/52) = 434 nm (blue)
Balmer-delta:
λ = 91.1/(1/4 - 1/62) = 410 nm (violet)
Recall:
Color Wavelength
violet 380–450 nm
blue 450–475 nm
cyan 476–495 nm
green 495–570 nm
yellow 570–590 nm
orange 590–620 nm
red 620–750 nm
3. Paschen series: → nf = 3
Paschen series are the series of transitions for an atom
when one of its electrons goes from a layer n to the layer
n = 3.
The transitions:
from n = 4 to n = 3 is called Paschen-alpha,
from n = 5 to n = 3 is Paschen-beta,
6 to 3 is Paschen-gamma, etc.
All these transitions are in the band of
the infrared.
We have the Rydberg formula:
λ = (1/Ry) . 1/(1/32 - 1/ni2)
= 91.1 /(1/32 - 1/n2) nm
Paschen-alpha:
λ (3) = 91.1/(1/9 - 1/42) = 1874 nm
Paschen-beta:
λ = 91.1/(1/9 - 1/52) = 1281 nm
Paschen-gamma:
λ = 91.1/(1/9 - 1/62) = 1093 nm
4. Brackett series:→ nf = 4
Brackett series are the series of transitions for an atom
when one of its electrons goes from a layer n to the layer
n = 4.
The transitions:
from n = 5 to n = 4 is called Brackett-alpha,
from n = 6 to n = 4 is Brackett-beta, etc.
All these transitions are in the band of
We have the Rydberg formula:
λ = (1/Ry) . 1/(1/42 - 1/ni2)
= 91.1 /(1/42 - 1/n2) nm
Paschen-alpha:
λ (4) = 91.1/(1/16 - 1/52) = 4050 nm
Paschen-beta:
λ = 91.1/(1/16 - 1/62) = 2624 nm
|
|